DEPARTMENT OF HEALTH & HUMAN SERVICES Intuitive Spine, LLC % Thogus Products Company / RP+M Ms. Jennifer Palinchik 33490
Millennium Dental Technologies Inc. Dr. David Harris Chief Science Officer I 0945 South Street Suite I 04-A Cerritos, CA 90703 R

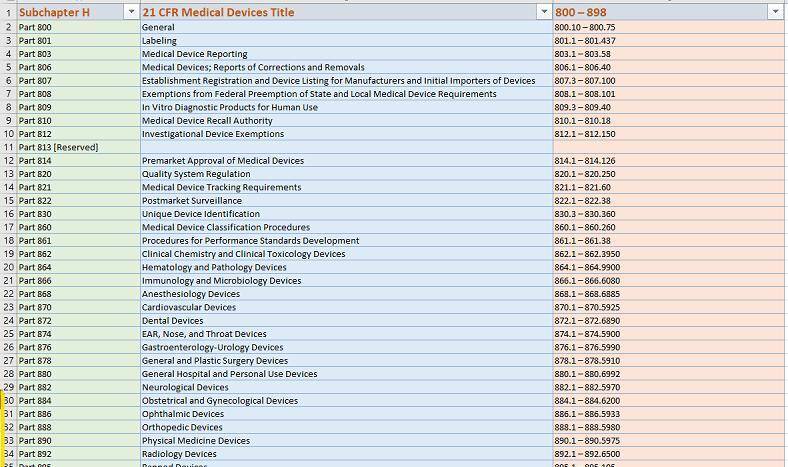
Code of Federal Regulations Title 21 Parts 800 to 1299 Food and Drugs: US Government Publishing Office, Office of the Federal Register: 9781683880820: Amazon.com: Books
.jpg)